CRAMPING in Skiing
By Kurt Jepson
Summer temperatures are not far away and those scheduled intensity sessions in heat and humidity will tax you mentally and physically. Athletes and coaches are aware of lessening the likelihood of muscle cramps by practicing sound hydration and electrolyte replacement strategies. But, is this true?
A deep drive into the available literature on Exercise Associated Muscle Cramps (EAMC) provides surprising information on the likely causes and remedies for this common athletic malady.
Exercise related muscle cramps are typically an unpredictable, intense, isolated, and temporary involuntary contraction of skeletal muscle fibers occurring during, or post, demanding physical activity. They are often associated with warmer environments.
Exercise associated muscle cramps (EAMC) routinely afflict “two joint muscles,” specifically those that act or cross 2 joints. Examples are the hamstrings (hip and knee), and calf (knee and ankle) muscles.
Cause and remedy have been debated for over 100 years and predictably, much “folklore” has developed around the topic of muscle cramping. The common nomenclature used to describe cramps is misleading. For example, terminology such as” heat cramps,” infers EAMC are specifically related to ambient temperatures and are negated via application of cooling modalities, neither of which is the case.
A $6.5 billion industry, as valued by Fortune Business Insight in 2021, has developed since Gatorade was introduced in 1965 at the University of Florida. Various products have been marketed in part, by the inference that “sports” beverages, or Oral Rehydration Solutions (ORS), provide prophylaxis against EAMC, particularly when exercising in hot and humid conditions.
Unfortunately, the literature falls short verifying this marketing concept. Most research provides predominantly observational and anecdotal data, leaving the athlete with limited sound scientific information regarding use or efficacy.

Anon, in the British Medical Journal also described “water poisoning” in 1923. Mistakenly, so much water was administered to cramping workers in hot environments, that sodium and chloride losses through sweating, lead to “Hyponatremia” (severe electrolyte depletion) and generalized muscle cramping ensued, not just those muscles involved in physical work. Although this science is outdated, and the methodology utilized was limited via the historic period, the concepts therein have not been fully ignored by modern scientists (Whitfield AHN. Too much of a good thing? The danger of water intoxication in endurance sports. Br J of General Prac. July 2006).
Effective prevention and remedy of any physiologic dysfunction begins with a sound knowledge of causation. Currently there are two major hypotheses of the etiology of EAMC.
A retrospective review of numerous studies from 1955 to 2008 by Miller and Stone, et al identified; the “Neuromuscular Based”, and the “Hydration-Electrolyte Depletion-Based” origins as most frequently named (Exercise-Associated Muscle Cramps: Causes, Treatment, and Prevention. Sports Health vol 2(4), 2010).
Let’s take a look at the merits and flaws of both theories.
Most studies that link cramping and dehydration and/or electrolyte depletion to EMAC, do so via Level 4 or 5 science, in other words, low scientific value.


More recent works have identified a possible connection between sweat rates in athletes and the incidence of EAMC.
A study by Bergeron et al in 2003, looked at 17 tennis players participating “in heat” and found that those with “a cramping history” had elevated sweat rates (~2,5 L/hr.) and higher sodium depletion rates (~2.7 g/hr.), than did non cramping individuals. A similar study looked at American Football players (n=5) exercising “in heat” and found that loss of sodium at > 1.18 g/hr. or sodium chloride at > 2,3 g/hr., resulted in a 9-fold increase in cramping incidence (Miller et al, Int J Sports Nutr Exer Metab. 2020). Ohno et al in 2018, documented the incidence of hamstring cramps that could be volitionally induced in subjects (n=9) after “passively” losing 1%,2% or 3% of their body mass via sauna use. Three of the 9 subjects cramped after 2-3% body mass reduction, or dehydration state.
These studies are somewhat compelling, but involved very small and athletically specific sample sizes.
Contradicting work by Miller et al, found no similar correlation in the football or tennis player situation noted above, among athletes from 10 other sports (Sweat characteristics of cramp-prone and cramp-resistant athletes. Int J Sports Nutr Exerc Metab. 2020; 30(3)).
In a nutshell, the Dehydration and Electrolyte Imbalance Theory of EAMC origin, relates perfuse sweating and systemic fluid loss to a contracture of the muscular interstitial space (the area occupied by muscle fibers and fluid combined), thereby increasing the concentration of neurologically “excitatory” chemicals and direct mechanical pressure on motor-nerve terminals (Bergeron MF, 2008).
Critics argue that if “salty sweating” is a leading cause of EAMC, how does one explain the fluid dynamics concept of less fluid moving out of the interstitial space as plasma osmolarity decreases (Schwellnus Br J Sports Med.2009)? A seemingly valid question.
A prospective cohort study of 72 runners, recruited at a South African ultra- marathon (56k), under went pre- and post-race body weight testing, blood analysis and filled out a detailed questionnaire highlighting their past cramping experiences. Race related cramping affected 21 runners of the 45 that had a history of EAMC. Twenty-two (n=22) non-cramping runners comprised the control group. Data analysis post-race showed no differences in serum levels of electrolytes, plasma volume, hematocrit, or osmolarity at various sampling time frames up to 1-hour post-race. The “cramping runners” were actually better hydrated than the non-cramping group in terms of body weight loss (2.9% vs 3.6%).
The authors concluded there was no relationship between the clinical recovery of EAMC and changes in serum electrolyte concentrations, or hydration levels as measured, up to 60 min post event (Schwellnus et al. Serum electrolyte concentrations and hydration status are not associated with exercise associated muscle cramping in distance runners. Br J Sports Med 2004. 38).

Numerous other conflicting studies have found no variation in plasma volume, red blood cell volume, muscle membrane potential/resting excitability, lost body mass, or plasma electrolyte concentrations during athletic competition, between “crampers” and “non-crampers” (Schwellnus 2003, Maughan 1986, Summers 2014, Costill 1976).
Another concept not adequately addressed by the dehydration/electrolyte depletion theory is that, those conditions are systemic, yet EAMC`s are quite isolated. They are typically seen only in working muscles that are fatigued or injured.
Additionally, “cold” weather athletes also experience EAMC. Maughan (J Sports Sci. 1986) reported that 18% of marathoners still experienced EAMC when competing at 10-12deg C.

Schwellnus, in the British Journal of Sports Medicine (June, vol 43 (6), 2009), cited 18 case series studies and one small case-controlled study (n=10) which supported the “electrolyte depletion” and “dehydration” mechanisms for the onset of EAMC. Unfortunately, these studies were based on anecdotal and observational data. Additionally, he cited results from 4 prospective cohort studies which did not support a causative connection between EAMC and dehydration/depletion.
In contrast, the “Altered Neuromuscular Control Theory” is quickly gaining traction as the explanation for EAMC. Most supportive data relate to human and animal models, as well as epidemiological studies in “cramping athletes”. Studies typically represent Level 1 and 2 Science.
This hypothesis was proposed in 1997, and updated in 2009, by MP Schwellnus in the British Journal of Sports Medicine (43(6), 2009). It relates the predictability of exercise associated muscle cramps to; onset of muscle fatigue, past or recent injury, nutritional status and other intrinsic or extrinsic stressors (i.e., ambient temperature), which produce an imbalance between motor nerve excitatory and inhibitory activity.
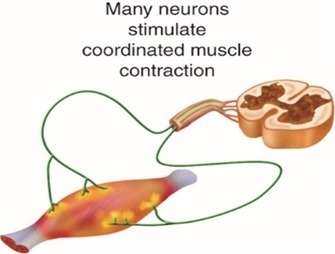
Animal studies have demonstrated (Hutton RS 1986, Nelson DL 1985) that fatigue alters the activity level of Muscle Spindle Fibers (MSF`s) which induce rapid muscle contractions and the Golgi Tendon Organs (GTO`s), which inhibit muscle contraction when activated.
Both mechanosensory receptors are protective by design. They bypass the motor cortex of the brain and as such, initiate an immediate contractile, or non-contractile, intramuscular response to potentially injurious elongation (MSF`s) or shortening (GTO`s).
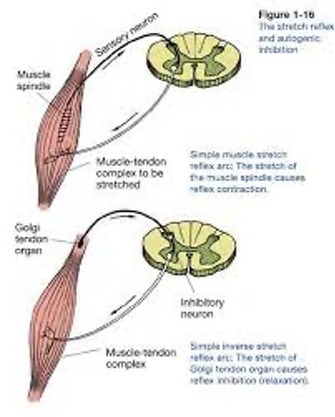
The GTO receptors likely play a leading role in EAMC. They reside in the musculotendinous junction of the contractile complex and are tension sensitive. When activated, they inhibit muscle contraction via Alpha Motor Nerve signal interruption, and the muscle “relaxes.” The Altered Neuromuscular Control Theory proposes that fatigue suppresses GTO response to “excessive” volitional or non-volitional contractions (which produce tendon tension) and left unchecked, this scenario quickly escalates to cramping.
The literature overwhelmingly points to the incidence of EAMC and fatigue induced alterations in neuromuscular function (Schwellnus 2008, 2010, Shang 2011, Stone 2010, Summers 2014, Maughan 1986).
Most available studies sample running athletes such as marathoners, ultra-marathoners, rugby and soccer players, triathletes, etc. Subject populations are often very large, for example, n=1300 marathoners (Manjra et al), n=16000 21k-56k runners (Schwellus et al) and n=41,698 distance runners (Swanevelder et al).
Fatigue related cramping predictors in these studies include; onset late in the contest or in a fatigued state, lack of training intensity or running experience leading up to the event, higher speed and work load than typically encountered, challenging terrain, nutritional status, heat and humidity, higher BMI, chronic disease state, age >40, and a family or individual history of EAMC.

Interestingly, pre- and post-race blood sampling in many of these works rarely indicant significant differences in serum electrolyte concentrations (Na, K, Mg, Cl, etc), hemoglobin levels, or plasma osmolarity between “crampers” and “non-crampers”. Hydration status via pre- and post-race body weight follows a similar trend.
It should be noted here, that sampling of plasma for electrolyte concentration does not likely give an accurate measure of what is actually going on with the muscle at the intracellular level, particularly at any given time interval. As mammals we strive to maintain metabolic balance, preserving intracellular ion concentrations even if extracellular plasma electrolytes become depleted, but homeostasis takes time (Costill DL, J Appl Physiol, 1976 and Sjogaard G, Acta Physiol Scand, 1986).
That said, serum analysis is the best test option available, versus complicated biopsy.
There is a lack of compelling evidence that EAMC onset is associated with electrolyte concentrations and hydration status. Similarly, there is very limited experimental data relating cessation of EAMC and the administration of electrolytes via Intravenous (IV) or ingestion avenues in the athletic population.
IV use at the “professional athlete” level is primarily a carryover from the general medical literature, where the connection between severe “global” skeletal muscle cramping and serum electrolyte disturbances has been well documented. These situations usually relate to medical conditions such as plasma volume contraction via vomiting and diarrhea, trauma, and patients undergoing hemodialysis (Knochel, Layzer, McGee, Neal). In those patients, IV intervention is very efficacious.
The Altered Neuromuscular Control theory of EAMC relating fatigue to alterations in muscle contraction checks many boxes versus the Dehydration-Electrolyte Depletion theory.

Another less discussed hypothesis for the origins of EAMC, relates to actual muscle damage.
Martinez-Navarro et al in 2020, published a study which looked at 98 marathon runners (Muscle Cramping in the Marathon: Dehydration and Electrolyte Depletion vs. Muscle Damage. J Strength Cond Res. Jun 1;36 (6)). They performed pre- and post-race cardiopulmonary testing, BMI, urine, and blood analysis. Twenty-four percent (24%) of the race finishers developed EAMC during or immediately after the race. Test results indicated no post-race differences in serum Na, K or other ion concentrations, no specific gravity variations in urine, and no body mass (BM) variations between the cramping and non-cramping groups. What was interesting, was that the “crampers” had significantly higher levels of post-race creatine kinase and lactate dehydrogenase. Both compounds are blood markers for muscle fiber damage, and this held true for the 24-hour sample as well. Additionally, the percentage of “crampers”, who by survey, participated in routine strength training as part of their usual fitness program, was only 25%, versus the “non-crampers” who participated in weight training at 47.6%.
It is unclear if weight training “re-biased” mechanoreceptor activity or simply added to muscle fiber durability. Either would hypothetically influence cramping. The obvious question is,….did cramping result from fiber damage due to insufficient conditioning in those individuals, or did the bio-markers appear in response to the vigorous muscle contraction associated with the act of cramping?
Either way, all things athletic benefit from gym time!

The last proposed mechanism for EAMC is perhaps the most interesting and difficult to study. O`Connell et al, did a retrospective genetic case-controlled study, analyzing 116 participants with a self-reported history of cramping related to ultra-endurance events. One hundred fifty individuals (n=150) with no history of EAMC were included as controls.
Individuals were genotyped for selected variants. The investigators found that a specific genotype (COL5A1 CC) was significantly overrepresented in the non-cramping group (21.3%) compared with the cramping group (11.1%). The authored looked at various other muscle tissue related genomes. There were no other overrepresented “polymorphisms” found in screened samples. This study implicated COL5A1 as a suspect marker for EAMC afflicted individuals (O`Connell K, et al. Collagen genes and Exercise-associated muscle cramping. Clin J Sports Med. 2013 Jan 23(1)).

Obviously, more work needs to be completed on this topic, but this could explain why some athletes are more prone to EAMC than others given similar challenging conditions.
It`s clear when examining the literature, EAMC very likely has a multifactorial origin, not unlike other maladies in human disease and dysfunction. To best address prevention and treatment then, multiple strategies should be employed.
Let’s look at some remedies that have enjoyed anecdotal success over the years, with scientific evidence in mind.
ATHLETE HISTORY;
Studies indicate those individuals with a history of EAMC are significantly more likely to have subsequent episodes when under training or competitive stress. Information on medical conditions, medication use, training load, environmental acclimatization, sleep habits, diet and hydration status, stimulant use (ie caffeine), and muscle injury history may all help predict EAMC.
These factors may affect an athlete’s Fatigue Threshold, produce neuro system over/under stimulation, influence nutritional input to muscle function, and enhance environmental physiologic stressors accelerating fatigue.
STRETCHING;
Stretching prophylactically fails to influence the incidence of EMAC (Miller et al. Muscle Nerve. 2018, 57(3)).
Stretching “acutely” for a cramping muscle is however, very effective in terms of relief and abatement of “spasm.” Self or administered passive stretching, has been shown to reduce electromyographic activity in the affected muscle. Proposed mechanisms focus on the tension-based response by the Golgi Tendon Organs leading to contractile inhibition (Bertolasi 1993, Schwellnus 2011, Khan 2007, Marquirriain 2007). The stretch may be applied as long as needed to inhibit muscle shortening, and often minutes of constant load are required, as are numerous repetitions.

EXERCISE AND TRAINING;
The documented cycle of events leading to EAMC usually involve some level of muscle fatigue. “Premature fatigue” is postulated to cause activation of muscles in a shortened length due to abbreviated range of motion (ie stride length, a contracted skiing posture, etc.), inhibition of GTO`s, decreased central nervous system modulation, and increased muscle spindle activity as eccentric control of fiber elongation lessens. All these components set the athlete up for involuntary muscle contraction.

The key to short circuiting these events is predicated by the delay of fatigue onset.
“Prevention” exercises for those prone to EAMC should involve neuromuscular education concepts, such as additional plyometric activities which enhance reflex sensitivity, nerve pathway efficiency and Central Nervous System (CNS) descending pathway activation. Descending CNS pathways temper or “smooth” muscle contractions (Chimera 2004, Swanik 2002, Wilk 1993).
This site (NTS) provides an endless supply of skier specific exercises.

Expanding endurance work will benefit the circulatory tissue response, increase plasma volume, and add to extracellular fluid levels, thus raising the fatigue threshold of the athlete (Hakkinen K et al, Fatiguability in voluntary and reflex contraction after conditioning of human skeletal muscle. Electromyogr Clin Neurophysiol, 1985;25).
Training at the temperatures, altitude, and intensities one expects to encounter at an upcoming competition, will also delay the onset of physiologic stress induced fatigue.
RECOVERY;
Rest and recovery strategies post “fun races” and training can not be ignored, even off season. Proper management of localized soft tissue injuries will also minimize adverse neuromuscular adaption that may predispose the athlete to EAMC down the road. All of the athletes` recovery tool box should be utilized; sleep, diet, soft tissue work, easy cross training, focused rehab, mental breaks, etc.
NUTRITION/HYDRATION;
Most of the studies which utilized oral rehydration solutions (ORS) to investigate their affect on EAMC, fail to use those solutions void of carbohydrates. Carbohydrate supplementation is a proven endurance performance enhancer and fatigue suppressant. A meta-analysis by Vandenbogaerde and Hopkins in 2011 (Sports Med. 41(9)), looked at 88 crossover studies in which carbohydrates, with and without protein, were utilized by athletes for strenuous activity. Substantial benefit was realized in terms of power output, time to exhaustion, and overall performance. Interestingly, the affect of “salt” was deemed “unexpectedly trivial.”
Jung et al at the University of Alabama, conducted a study where subjects (n=13) performed calf exercises to fatigue in a hot environment (dry blub temp of 37 deg C ). Trials included consumption of an ORS drink and a “hypohydration state”, or no fluid consumption allowed. Nine of the 13 participants (69%) experienced cramps in the ORS trial and only 7 of the 13 in the “no fluid allowed” trial. In the ORS trial however, they determined that the onset of cramping was significantly delayed ( ~36 min vs ~14 min) versus the non-drinking group.
An article in the Journal of the International Society of Sports Nutrition by Wing et al (Effect of oral rehydration solution versus spring water intake during exercise in the heat on muscle cramp susceptibility of young men. 2021. 18(22)), hypothesized that post-exercise cramping would be more prevalent in study participants (n=10) that consumed only spring water vs an ORS. The men ran downhill in 35-36 deg C temps for 40-60 minutes to reduce their body mass by 1.5-2%. They then underwent calf cramp “Threshold Frequency” testing (TF), utilizing electrical stimulation to induce EAMC at various time intervals post run. Related blood panels were also analyzed.
They found that ingestion of the ORS indeed raised the TF to cramping vs the spring water group. Also, the water group experienced osmolarity reduction and a serum ion concentration decrease of <2%. The authors concluded that ORS do have a beneficial effect on EAMC.
These studies exemplify the value of intra and pre-post event utilization of ORS in terms of maximizing performance, delaying muscle fatigue and therefore indirectly impeding EAMC.
A proactive approach to fluid intake and energy supplementation with those athletes susceptible to EAMC is prudent. The National Athletic Trainers Association (NATA) recommends an athlete’s body mass be monitored when exercising in challenging environments and that fluid management allows for no more than a 2% reduction in body weight. The American College of Sports Medicine (ACSM) recommends that athletes drink the maximum amount tolerated during a contest, or between 650-1200 ml per hour. The New York City Marathon medical director suggested participants ingest no more than 750 ml per hour in 2005. As you can see there is some disagreement with dosage among experts.
Common sense and fluid intake adjustment to the training environment is likely the answer. Plain water consumption is important to performance and recovery but don`t go overboard with water consumption.

FOLKLORE;
Because EAMC reportedly affects so many athletes, 67% of triathletes, 18-705 of marathoners or cyclists, and 30-53% of American Football players (Maughan,Miles, Manjra and others), an atmosphere for “folklore remedy” has flourished. Some popular strategies are listed below.
-Quinine/Tonic water.
A Cochrane review in 2015 reported findings from 23 clinical trials and 1586 participants suffering from muscle cramps of any cause and in any location, treated with quinine in isolation or as part of a mixture. The authors concluded that, “There is low quality evidence that quinine (200mg to 500mg per day) significantly reduces cramp number and cramp days and moderate evidence that quinine reduces cramp intensity.” Adverse side affects are categorized as “not significantly greater than placebo.” Gastrointestinal distress is common.
Most clinicians, including myself, do not recommend its use in isolation or as part of a compound, to treat EAMC.
Because death can occur, although very rare, some countries restrict the prescription of quinine.

-Pickle juice.
Miller et al completed a study where 1ml of pickle juice was ingested 2 sec after the initiation of a muscle cramp and found cramp duration was reduced 37% versus water (J Ath Train 2009). They commented that such a small amount of pickle juice would have no meaningful affect on plasma ion concentrations and the rapid response was not consistent with GI absorption.
They proposed that the activation of receptors in the mouth (oropharyngeal) via the harsh taste, resulted in a decreased firing rate of alpha motor nerves to the affected cramping muscle. Our nervous systems can only process so much information at once and prioritizes those sensations it interprets as “dangerous” first. Pickle juice “danger” may win out over cramping in this case.

The same mechanism as above. Caution,….these substances are VERY salty!
-Chili peppers, wasabi, ginger, capsaicin.
Similar to the above explanation, these substances can invoke a powerful stimulation response in the mouth. “Transient Receptor Potential” (TRP) theory and its effect on muscle cramps was developed by DH Craighead and published in Muscle Nerve in 2017. Although research is quite limited, the theory goes that TRP receptors form a family of 28 related ion channels which help mediate taste and pain in the mouth.
There is little doubt that unpleasant tastes in the mouth can induce certain electrical activity in the brain. There would seem to be a significant gap between that activity and inhibition of a motor nerve traveling to a distal muscle group. More investigation is needed.
-Bananas.
Potassium is not considered an electrolyte of interest in EAMC. One study showed that it took 60 min post ingestion for bananas to have an influence on plasma Potassium levels. It did take only 15 min for Glucose levels to rise (Miller. 2012).
Eat bananas as a fuel source, not as a direct combatant to cramping.

CONCLUSIONS;
Exercise associated muscle cramps (EAMC) are a prevalent, non-life threatening, performance interrupting, athletic malady of a mixed and complicated pathophysiologic etiology. Anecdotal information clouds the origins, identification, and management of EAMC.
One take away is clear, a compromised Fatigue Threshold (FT) leaves one exposed to EAMC. Manage cramping with a sound athletic multifactorial approach. Namely; train, eat, drink, rest. Sound familiar?
Get after it this summer!
