Physiological effects of Altitude - Part 2
Part 2: Introduction to altitude training (methods and advice)
By Sarah Willis, PhD
Many researchers have been studying to understand the stress of hypoxia (low level of oxygen that we have in higher altitude environments) as a stimulus and how we can best use it to adapt the body and increase performance while improving overall health.
We can live high (LH) and focus on the changes and adaptations our blood makes over time which allows for better oxygen delivery to our muscles, OR we can live low and train high (LLTH) and challenge the vascular system to increase the vasoreactivity of our vessels and allow better perfusion of oxygen into our tissues.
There are several combinations of methods, living high or low, while training high and/or low and the efficacy of these methods depends on several factors for proper execution including method of hypoxic stimulus and severity of hypoxia, duration of time exposed (hypoxic dose), intensity of exercise, etc.
A comprehensive panorama of the numerous training methods using different forms and protocols of hypoxia is shown below (Millet et al., 2013a; Girard et al., 2017). This article will focus first on the blood adaptations of living in altitude environments and later discuss hypoxic training methods to adapt the vasculature in efficiently utilizing oxygen while living in lower elevations or at sea-level.
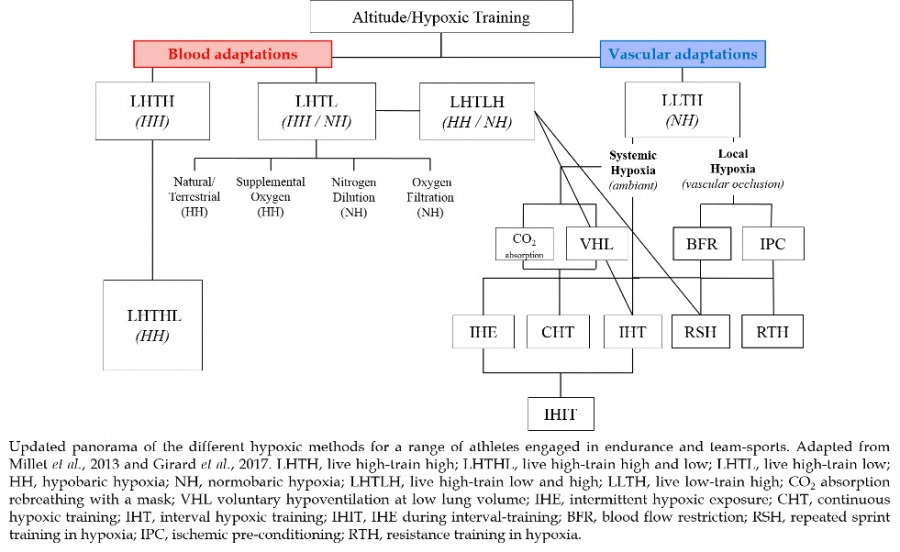
Live High and Train Low…how does it work?
Traditionally, athletes attend altitude training camps where they both live and train at moderate altitude (1800-2500 m, LHTH) for typically 2-4 weeks, 2-3 times a year (Wilber, 2007; Millet et al., 2010). This method involves generous time and financial commitment while also brings questionable sea-level performance benefits as there are large variations in individual responses (McLean et al., 2013). The well-known, live high-train low (LHTL) philosophy is considered as the ‘Gold-standard’ method for altitude training (Levine and Stray-Gundersen, 1997), though it is debated for its efficacy in elite endurance athletes (Siebenmann et al., 2012), underlying mechanisms (Gore and Hopkins, 2005; Levine and Stray-Gundersen, 2005), and the degree and type of the hypoxic stimulus (hypobaric vs normobaric) (Millet et al., 2012a; 2013b). There is a difference between 'real' altitude (hypobaric hypoxia) and simulated altitude (normobaric hypoxia) and research suggests that simulated altitude is not quite as effective as real altitude (Saugy et al., 2016). But, in any case, the simulation of altitude can be quite beneficial for athletes. This type of altitude training intervention is based on the principle that chronic exposure to an altitude environment stresses the body to make adaptations in your blood, increasing the erythropoietic effect (production of more red blood cells) and hemoglobin mass (oxygen carrying molecule in the blood) and thus allows the blood to deliver more oxygen to working muscles (Wehrlin et al., 2006). It has been well documented that hemoglobin mass increases at approximately 1% for every 100 hours of altitude exposure regardless of the exposure method, meaning living in real (hypobaric) or simulated (normobaric) hypoxia and whether training was performed in hypoxia or normoxia [i.e., live high train high, LHTH (> 2100 m) or live high train low, LHTL (~3000 m)] (Gore et al., 2013).
An important consideration of these training methods is the altitude dose (how much exposure is needed), as it has been suggested that reaching an altitude of 2200-2500 m for at least 4 weeks with ideally 20-22 hours of hypoxic exposure/day is optimal for improved hematological changes and sea-level performance where longer exposure is better (Millet et al., 2010). It has been shown that benefits still occur in the blood adaptation while spending ideally 16 hours/day and minimum 12 hours/day (Millet et al., 2010) (especially important when considering sleeping in an altitude tent). When using simulated altitude (chamber, sleeping tent, hypoxic generator system for training, etc.), it is generally recommended to use slightly higher altitudes to better stimulate similarly to real altitude.
We need to be careful of how your body reacts to this additional stress of the altitude environment. I recommend monitoring your body with oxygen saturation (fingertip measurement) and morning heart rate recovery testing (See Physiological Effects of Altitude Part 1). This is especially important when using altitude training methods (particularly living high), as you should still be able to perform your training as suggested by your coach/team. Sleep is of extreme importance - make sure you get extra sleep, longer at night, or take a short nap in the afternoon if possible. It is also important to hydrate and eat well to maintain your capacities and help your body handle the extra stress of altitude exposure. Special attention should be given to your training upon return to sea-level after an altitude training camp, as the body needs time to recover from the extra stimulus of altitude exposure and training and fatigue levels should be monitored closely to optimize the performance benefits upon return to sea-level competition (Chapman et al., 2014).
Living low and train high, how does it work?
While living at altitude elicits hematological adaptations to hypoxia and allows for subsequential improvement of oxygen supply to the tissues, living low and training in hypoxia (LLTH) challenges the efficiency of the cardiovascular system to transport and perfuse the muscles with oxygenated blood to stimulate vessel adaptation and improve performance (see above panorama of altitude training methods). Even though LLTH studies have reported improved oxygen utilization within the muscle (Vogt et al., 2001; Ponsot et al., 2006; Zoll et al., 2006) through advantages for aerobic and anaerobic performance, which depends on the exercise mode, duration, and intensity, the amount of hypoxic exposure is inconclusive (Millet et al., 2010). Furthermore, it has been demonstrated that athletic performance is likely improved with intermittent hypoxic training (IHT) at high intensity by increased mitochondrial efficiency and improved pH/lactate regulation (Katayama et al., 2003; Ponsot et al., 2006; Zoll et al., 2006).
The physiological challenge associated with exercise in hypoxia is evident as mentioned above. However, the additional influence of high-intensity exercise must be considered as there are several advantageous adaptations to the cardiovascular and muscle systems that occur including: increased skeletal muscle oxidative capacity, increased resting glycogen content, increased capacity for muscle lipid oxidation, enhanced peripheral vascular structure and function, and improved time-trial and maximal performance test results (Burgomaster et al., 2005; Gibala et al., 2006; Burgomaster et al., 2008; Rakobowchuk et al., 2008). Generally, exercise leads to increases in blood flow and thereby increases laminar shear stress in the vessel, which leading to upregulation of endothelial nitric oxide synthase (an enzyme which allows smooth muscle relaxation, vasodilation, and increased blood flow) (Gielen et al., 2001; Tinken et al., 2009). This response is proportional to exercise intensity and therefore, the demand should increase with high-intensity exercise (such as repeated sprint performance) as previously suggested (Mortensen et al., 2008; Casey and Joyner, 2012). Similar vasodilation mechanisms exist during conditions of systemic hypoxia (at altitude) but to a greater extent due to the reduced oxygen availability (Casey and Joyner, 2012).
Combining the metabolic stress of high-intensity interval exercise with a hypoxic stimulus should induce an effective stimulus to prompt physiological responses and spur biological reactions.
Repeated Sprint Training in Hypoxia (RSH)
An innovative training method was developed involving demanding repeated bursts of high intensity exercise (≤ 30s) in hypoxia (repeated sprint training in hypoxia, RSH) to maximize the metabolic stress and has demonstrated improved maximal performance and delayed fatigue when compared to the same training but in normoxia (RSN) (Faiss et al., 2013). Performing this repeated high-intensity sprint exercise in hypoxia places a great physiological and metabolic demand on the body in short periods of time with primarily reliance on anaerobic glycolysis and a shift towards aerobic energy metabolism as sprinting is repeated (Gaitanos et al., 1993; Bogdanis et al., 1995). This then allowed the combination of the known principles of LHTL methods with RSH, meaning living high while training low while including sessions of the RSH intervention (LHTLH). Among the specific responses associated to LHTLH, there are increased mRNA levels involved in oxygen signaling, oxygen transport, mitochondrial biogenesis, and metabolism (Brocherie et al., 2018), as well as positive functional adaptations as shown by the improved oxidative capacity in type I and type II fibers, while maintaining fiber size (van der Zwaard et al., 2018). From these investigations, it becomes clear that high intensity training provides an important metabolic stress when combined with hypoxia to stimulate peripheral adaptations.
Since 2013, repeated sprint training in hypoxia has been successful across several exercise modes [leg cycling (Faiss et al., 2013), cross-country skiing (Faiss, Willis et al., 2014), running (Galvin et al., 2013), and team sports (Brocherie et al., 2014), with a meta-analysis (Brocherie et al., 2017)], demonstrating an increase in power output and number of sprints. It has been indicated that RSH is a beneficial mode of altitude/hypoxic training demonstrating improvements in performance (Millet et al., 2013b).
This has led to new methods which combine repeated sprint training with novel ways of creating a hypoxic environment either systemically (whole body exposure to lower oxygen environment) or locally (creating a hypoxic environment by limiting blood flow to tissues, i.e., blood flow restriction).
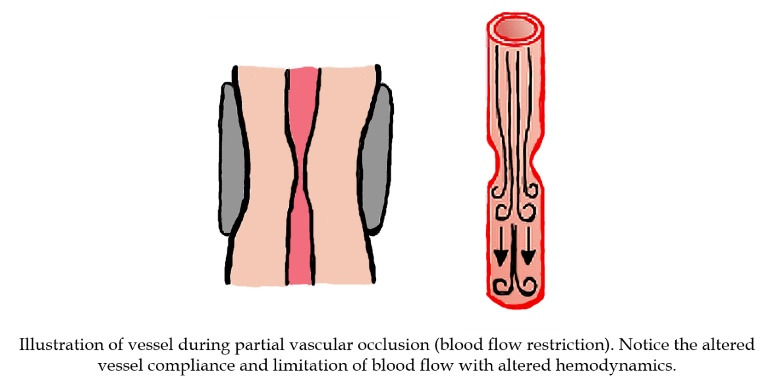
Local hypoxia via blood flow restriction (BFR)
It has been shown that severe hypoxia can be created locally in the muscle tissue (ischemia) by applying a blood flow restriction (BFR, also known as vascular occlusion) to the limbs during exercise (Scott et al., 2014). This external application of pressure sufficiently allows the maintenance of arterial blood inflow (partial vascular occlusion as a percentage of resting total occlusion pressure) while completely (or almost completely) occluding the venous outflow (Kaijser et al., 1990), thus increasing blood in the tissues distal (away from the heart) to the occlusion.
When compared to systemic hypoxia, using BFR that induces both ischemia and local hypoxia likely has a different effect on vascular/endothelial function due to specific vascular resistance and vessel diameter adjustments, accumulation of metabolites (nitric oxide, adenosine, prostaglandins, hydrogen ions, etc.) and/or indirect sympathetic activation. In these BFR conditions with restricted vasodilation, the demand for increased blood flow cannot be met with similar mechanisms as during systemic hypoxia (hypoxia-induced vasodilation). Thus, there are both different methods (systemic, via lower fraction of inspired oxygen; and local, via greater vessel resistance and lower blood flow) and intrinsic mechanisms (systemic, metabolic vasodilation to increase blood flow for oxygen delivery, and local, greater vascular challenge for blood flow regulation) by which a hypoxic environment may exist, and in which alterations of vascular conductance and blood flow are present.
My recent research has combined repeated sprint exercise with blood flow restriction and demonstrated that the vascular resistance in the vessel due to external pressure of the cuff reduces oxygen consumption and therefore performance (showing that local hypoxia indeed limits oxygen delivery) (Willis et al., 2018).
We suggested that BFR may be a similar or additional stimulus to hypoxia to elicit greater blood volume and perfusion to the tissue, stimulating vascular adaptations (Willis et al., 2019a, b). Our results have suggested that arms may have greater vascular reactivity than the legs regarding the sensitivity to blood volume likely due to the oxygen transport and vascular properties of arms versus legs (Willis et al., 2019a). Oxygenation responses vary with exercise intensity likely related to the reliance on different energy metabolism pathways, which also affect limbs differently.
In terms of performance, this means that BFR is a way to challenge vascular adaptation similar to hypoxia and may bring an additional hypertrophic effect which is being analyzed in training studies focusing on the molecular and hormonal adaptations. Additionally, we have identified that arms have greater vascular sensitivity, therefore we should consider different training protocols between arms and legs.
Effects on performance
Implementing these methods into training programs is likely beneficial for increasing the capacity and reactivity of blood and oxygen transport, possibly improving the diffusion capacity along with vessel adaptations which may altogether enhance endothelial function leading to improved tissue oxygenation and performance. The implementation of short training blocks with these methods can be valuable to induce a rapid and robust stimulus in a short time frame, thus providing a time-saving strategy and effective method for adaptations to occur. It is interesting to pursue this training across different sports, as this type of repeated sprint training may be applicable for not only team-sport athletes, but also endurance athletes (i.e., pacing surges and increases in tempo). The concept of using BFR in training can provide a greater stimulus and an increased workload while presenting a new stimulus for adaptations. Different training programs should be designed with special attention placed on limb differences, as arms seem to have a greater and more reactive vascular response than legs.
Specific training recommendations
While the optimal stimulus depends on the population of interest, suggested training recommendations are as follows and supported by a recent meta-analysis (Brocherie et al., 2017). The high-intensity exercise protocol (whether in hypoxia or normoxia and whether with or without BFR) should last approximately 60 min including the warm-up and cool-down periods and be performed 2-3 times a week during blocks of training (2-5 weeks duration as part of a periodized training program. During each session, the athlete performs a series of 3-4 sets of 4-7 maximal ‘all-out’ sprints (4-15 s duration) in hypoxia (3000-3800 m and 14.2-12.8% FiO2) and/or with BFR of about 45% of total occlusion pressure. It is important to achieve a specific sprint-to-rest ratio of 1:2 to 1:4 with inter-set recovery about 3-5 min without occlusion. This specific training should be performed in alteration with low-intensity exercise to maintain and develop basic fitness along with aerobic metabolism.

This article was written as an introduction to several altitude training methods. If you have questions or specific inquiries, please email me at sarah.jean.willis.15@gmail.com for consultation, advice, and project planning.
References
Bogdanis, G.C., Nevill, M.E., Boobis, L.H., Lakomy, H.K., and Nevill, A.M. (1995). Recovery of power output and muscle metabolites following 30 s of maximal sprint cycling in man. J Physiol 482 ( Pt 2), 467-480.
Brocherie, F., Millet, G.P., and Girard, O. (2014). Neuro-mechanical and metabolic adjustments to the repeated anaerobic sprint test in professional football players. Eur J Appl Physiol. doi: 10.1007/s00421-014-3070-z.
Brocherie, F., Girard, O., Faiss, R., and Millet, G.P. (2017). Effects of Repeated-Sprint Training in Hypoxia on Sea-Level Performance: A Meta-Analysis. Sports Med 47(8), 1651-1660. doi: 10.1007/s40279-017-0685-3.
Brocherie, F., Millet, G.P., D'Hulst, G., Van Thienen, R., Deldicque, L., and Girard, O. (2018). Repeated maximal-intensity hypoxic exercise superimposed to hypoxic residence boosts skeletal muscle transcriptional responses in elite team-sport athletes. Acta Physiol (Oxf) 222(1). doi: 10.1111/apha.12851.
Burgomaster, K.A., Hughes, S.C., Heigenhauser, G.J., Bradwell, S.N., and Gibala, M.J. (2005). Six sessions of sprint interval training increases muscle oxidative potential and cycle endurance capacity in humans. J Appl Physiol (1985) 98(6), 1985-1990. doi: 10.1152/japplphysiol.01095.2004.
Burgomaster, K.A., Howarth, K.R., Phillips, S.M., Rakobowchuk, M., Macdonald, M.J., McGee, S.L., et al. (2008). Similar metabolic adaptations during exercise after low volume sprint interval and traditional endurance training in humans. J Physiol 586(1), 151-160. doi: 10.1113/jphysiol.2007.142109.
Casey, D.P., and Joyner, M.J. (2012). Compensatory vasodilatation during hypoxic exercise: mechanisms responsible for matching oxygen supply to demand. J Physiol 590(24), 6321-6326. doi: 10.1113/jphysiol.2012.242396.
Chapman, R.F., Laymon Stickford, A.S., Lundby, C., Levine, B. (2014). Timing of return from altitude training for optimal sea level performance. J Appl Physiol 116: 837-843.
Faiss, R., Leger, B., Vesin, J.M., Fournier, P.E., Eggel, Y., Deriaz, O., et al. (2013). Significant molecular and systemic adaptations after repeated sprint training in hypoxia. PLoS One 8(2), e56522. doi: 10.1371/journal.pone.0056522.
Faiss, R., Willis, S., Born, D.P., Sperlich, B., Vesin, J.M., Holmberg, H.C., et al. (2014). Repeated Double-Poling Sprint Training in Hypoxia by Competitive Cross-country Skiers. Med Sci Sports Exerc. doi: 10.1249/MSS.0000000000000464.
Gaitanos, G.C., Williams, C., Boobis, L.H., and Brooks, S. (1993). Human muscle metabolism during intermittent maximal exercise. J Appl Physiol (1985) 75(2), 712-719.
Galvin, H.M., Cooke, K., Sumners, D.P., Mileva, K.N., and Bowtell, J.L. (2013). Repeated sprint training in normobaric hypoxia. Br J Sports Med 47 Suppl 1, i74-79. doi: 10.1136/bjsports-2013-092826.
Gibala, M.J., Little, J.P., van Essen, M., Wilkin, G.P., Burgomaster, K.A., Safdar, A., et al. (2006). Short-term sprint interval versus traditional endurance training: similar initial adaptations in human skeletal muscle and exercise performance. J Physiol 575(Pt 3), 901-911. doi: 10.1113/jphysiol.2006.112094.
Gielen, S., Schuler, G., and Hambrecht, R. (2001). Exercise training in coronary artery disease and coronary vasomotion. Circulation 103(1), E1-6.
Girard, O., Brocherie, F., and Millet, G.P. (2017). Effects of Altitude/Hypoxia on Single- and Multiple-Sprint Performance: A Comprehensive Review. Sports Med. doi: 10.1007/s40279-017-0733-z.
Gore, C.J., and Hopkins, W.G. (2005). Counterpoint: positive effects of intermittent hypoxia (live high:train low) on exercise performance are not mediated primarily by augmented red cell volume. J Appl Physiol (1985) 99(5),2055-2057; discussion 2057-2058. doi: 10.1152/japplphysiol.00820.2005.
Gore, C.J., Sharpe, K., Garvican-Lewis, L.A., Saunders, P.U., Humberstone, C.E., Robertson, E.Y., et al. (2013). Altitude training and haemoglobin mass from the optimised carbon monoxide rebreathing method determined by a meta-analysis. Br J Sports Med 47 Suppl 1, i31-39. doi: 10.1136/bjsports-2013-092840.
Kaijser, L., Sundberg, C.J., Eiken, O., Nygren, A., Esbjornsson, M., Sylven, C., et al. (1990). Muscle oxidative capacity and work performance after training under local leg ischemia. J Appl Physiol (1985) 69(2), 785-787.
Katayama, K., Matsuo, H., Ishida, K., Mori, S., and Miyamura, M. (2003). Intermittent hypoxia improves endurance performance and submaximal exercise efficiency. High Alt Med Biol 4(3), 291-304. doi: 10.1089/152702903769192250.
Levine, B.D., and Stray-Gundersen, J. (1997). "Living high-training low": effect of moderate-altitude acclimatization with low-altitude training on performance. J Appl Physiol (1985) 83(1), 102-112. doi: 10.1152/jappl.1997.83.1.102.
Levine, B.D., and Stray-Gundersen, J. (2005). Point: positive effects of intermittent hypoxia (live high:train low) on exercise performance are mediated primarily by augmented red cell volume. J Appl Physiol (1985) 99(5),2053-2055. doi: 10.1152/japplphysiol.00877.2005.
McLean, B.D., Buttifant, D., Gore, C.J., White, K., Liess, C., and Kemp, J. (2013). Physiological and performance responses to a preseason altitude-training camp in elite team-sport athletes. Int J Sports Physiol Perform8(4), 391-399.
Millet, G.P., Roels, B., Schmitt, L., Woorons, X., and Richalet, J.P. (2010). Combining hypoxic methods for peak performance. Sports Med 40(1), 1-25. doi: 10.2165/11317920-000000000-00000.
Millet, G.P., Faiss, R., and Pialoux, V. (2012a). Point: Hypobaric hypoxia induces different physiological responses from normobaric hypoxia. J Appl Physiol (1985) 112(10), 1783-1784. doi: 10.1152/japplphysiol.00067.2012.
Millet, G.P., Faiss, R., Brocherie, F., and Girard, O. (2013a). Hypoxic training and team sports: a challenge to traditional methods? Br J Sports Med 47 Suppl 1, i6-7. doi: 10.1136/bjsports-2013-092793.
Millet, G.P., Faiss, R., and Pialoux, V. (2013b). Evidence for differences between hypobaric and normobaric hypoxia is conclusive. Exerc Sport Sci Rev 41(2), 133. doi: 10.1097/JES.0b013e318271a5e1.
Mortensen, S.P., Damsgaard, R., Dawson, E.A., Secher, N.H., and Gonzalez-Alonso, J. (2008). Restrictions in systemic and locomotor skeletal muscle perfusion, oxygen supply and VO2 during high-intensity whole-body exercise in humans. J Physiol 586(10), 2621-2635. doi: 10.1113/jphysiol.2007.149401.
Ponsot, E., Dufour, S.P., Zoll, J., Doutrelau, S., N'Guessan, B., Geny, B., et al. (2006). Exercise training in normobaric hypoxia in endurance runners. II. Improvement of mitochondrial properties in skeletal muscle. J Appl Physiol (1985) 100(4), 1249-1257. doi: 10.1152/japplphysiol.00361.2005.
Rakobowchuk, M., Tanguay, S., Burgomaster, K.A., Howarth, K.R., Gibala, M.J., and MacDonald, M.J. (2008). Sprint interval and traditional endurance training induce similar improvements in peripheral arterial stiffness and flow-mediated dilation in healthy humans. Am J Physiol Regul Integr Comp Physiol 295(1), R236-242. doi: 10.1152/ajpregu.00069.2008.
Saugy, J.J., Schmitt, L., Hauser, A., Constantin, G., Cejuela, R., Faiss, R., Wehrlin, J.P., Rosset, J., Robinson, N., Millet, G.P. (2016). Same performance changes after live high-train low in normobaric vs. hypobaric hypoxia. Front Physiol 7:138.
Scott, B.R., Slattery, K.M., Sculley, D.V., and Dascombe, B.J. (2014). Hypoxia and resistance exercise: a comparison of localized and systemic methods. Sports Med 44(8), 1037-1054. doi: 10.1007/s40279-014-0177-7.
Siebenmann, C., Robach, P., Jacobs, R.A., Rasmussen, P., Nordsborg, N., Diaz, V., et al. (2012). "Live high-train low" using normobaric hypoxia: a double-blinded, placebo-controlled study. J Appl Physiol (1985) 112(1),106-117. doi: 10.1152/japplphysiol.00388.2011.
Tinken, T.M., Thijssen, D.H., Hopkins, N., Black, M.A., Dawson, E.A., Minson, C.T., et al. (2009). Impact of shear rate modulation on vascular function in humans. Hypertension 54(2), 278-285. doi: 10.1161/HYPERTENSIONAHA.109.134361.
van der Zwaard, S., Brocherie, F., Kom, B.L.G., Millet, G.P., Deldicque, L., van der Laarse, W.J., et al. (2018). Adaptations in muscle oxidative capacity, fiber size, and oxygen supply capacity after repeated-sprint training in hypoxia combined with chronic hypoxic exposure. J Appl Physiol (1985) 124(6), 1403-1412. doi: 10.1152/japplphysiol.00946.2017.
Vogt, M., Puntschart, A., Geiser, J., Zuleger, C., Billeter, R., and Hoppeler, H. (2001). Molecular adaptations in human skeletal muscle to endurance training under simulated hypoxic conditions. J Appl Physiol (1985)91(1), 173-182. doi: 10.1152/jappl.2001.91.1.173.
Wehrlin, J.P., Zuest, P., Hallen, J., and Marti, B. (2006). Live high-train low for 24 days increases hemoglobin mass and red cell volume in elite endurance athletes. J Appl Physiol (1985) 100(6), 1938-1945. doi: 10.1152/japplphysiol.01284.2005.
Wilber, R.L. (2007). Application of altitude/hypoxic training by elite athletes. Med Sci Sports Exerc 39(9), 1610-1624. doi: 10.1249/mss.0b013e3180de49e6.
Willis, S.J., Alvarez, L., Borrani, F., and Millet, G.P. (2018). Oxygenation time course and neuromuscular fatigue during repeated cycling sprints with bilateral blood flow restriction. Physiol Rep 6(19), e13872. doi: 10.14814/phy2.13872.
Willis, S.J., Borrani, F., and Millet, G.P. (2019a). Leg- vs arm-cycling repeated sprints with blood flow restriction and systemic hypoxia. Eur J Appl Physiol 119(8), 1819-1828. doi: 10.1007/s00421-019-04171-0.
Willis, S.J., Peyrard, A., Rupp, T., Borrani, F., and Millet, G.P. (2019b). Vascular and oxygenation responses of local ischemia and systemic hypoxia during arm cycling repeated sprints. J Sci Med Sport. doi: 10.1016/j.jsams.2019.05.001.
Zoll, J., Ponsot, E., Dufour, S., Doutreleau, S., Ventura-Clapier, R., Vogt, M., et al. (2006). Exercise training in normobaric hypoxia in endurance runners. III. Muscular adjustments of selected gene transcripts. J Appl Physiol (1985) 100(4), 1258-1266. doi: 10.1152/japplphysiol.00359.2005.
